Biohaven Highlights Progress Across Innovative Portfolio and Outlines 2024 Anticipated Milestones at the 42nd Annual J.P. Morgan Healthcare Conference; Established Extensive Portfolio Across 20 Therapeutic Indications in Neuroscience, Immunology and Oncology
- Five proprietary platforms fueling multiple clinical programs
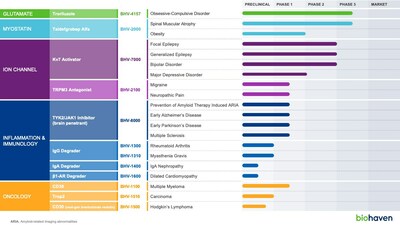
- Targeting large indications including epilepsy, bipolar disorder, depression, obsessive-compulsive disorder, migraine, pain, obesity, Alzheimer's disease, Parkinson's disease, multiple sclerosis, rheumatoid arthritis, and cancer. Plus, rare autoimmune and inflammatory diseases, including myasthenia gravis, cardiomyopathy, spinal muscular atrophy and IgA nephropathy
Further, our innovative extracellular protein degrader platform has generated multiple new investigational agents that are rapidly advancing into the clinic, initially targeting IgG, IgA, and β1-AR autoantibodies to treat both common and rare autoimmune diseases. This highlights the uniqueness of the platform to efficiently deliver highly differentiated assets that are finely tuned to specific clinical targets in relatively short development periods. For example, our newly disclosed β1-AR degrader went from concept to lead drug candidate in approximately one year. Our lead degrader candidate for β1-AR autoantibodies, BHV-1600, offers to re-route pathogenic antibodies to the liver where they can be degraded and help alleviate β1-AR+ heart failure. In addition, we have quickly advanced our next generation IgG degrader, BHV-1310, and shown that it can achieve ultra-rapid 90% IgG depletion after a single dose in preclinical models. This is particularly well suited to address indications such as acute myasthenia gravis, transplant rejection and other indications that require rapid clearance of disease-causing antibodies. In addition to completing enrollment in a pivotal spinal muscular atrophy clinical study, we have been opportunistic in exploring our myostatin inhibitor, taldefgrobep alfa, as a potential treatment approach for obesity demonstrating that it reduces adipose tissue and improves lean mass. Addressing the growing public health crisis of obesity by reducing fat while preserving and improving muscle mass would be a tremendous advancement in the field, especially as the field evaluates the potential long-term impact of reducing muscle mass that has been observed with GLP-1 medications. Finally, we look forward to an important milestone in our glutamate modulating program with the first quarter database lock for our interim efficacy analysis of our Phase 3 study of troriluzole in OCD. There have been no novel treatments in OCD in over 20 years and if troriluzole proves to be efficacious this will be an important breakthrough for the millions of patients suffering from this disorder."
Anticipated 2024 Clinical Milestones
Biohaven is positioned to achieve significant, value-creating milestones in 2024 across numerous programs:
Selective Kv7 Activator: BHV-7000 is a selective activator of Kv7.2/7.3 potassium channels, a breakthrough target in neurology and neuropsychiatry with blockbuster potential. Kv7 activation is a clinically validated target for treating epilepsy. More recently, clinical proof-of-concept studies have also demonstrated that Kv7 activators have robust antidepressant effects and rapid onset of action, providing strong clinical support for the transformative potential of BHV-7000 as a novel treatment for major depressive disorder.
- Initiate BHV-7000 Phase 2/3 program in focal epilepsy in 1Q 2024
- Initiate BHV-7000 Phase 2/3 study in bipolar disorder in 1Q 2024
- Initiate BHV-7000 Phase 2 study in major depressive disorder in 1Q 2024
- Initiate BHV-7000 Phase 2/3 study in generalized epilepsy in 2Q 2024
Troriluzole: Troriluzole is a novel glutamate modulator currently in Phase 3 development for obsessive-compulsive disorder (OCD). It is being evaluated as an adjunctive therapy in patients with an inadequate response to existing standard of care treatment. The troriluzole Phase 2 trial in OCD demonstrated consistent numerical benefits vs. placebo on the Yale-Brown Obsessive Compulsive Scale (primary endpoint) at all timepoints and informed the Phase 3 study design.
- Database lock in 1Q 2024 and report troriluzole Phase 3 interim efficacy analysis topline results in OCD in 2Q 2024
Taldefgrobep alfa: Taldefgrobep is a novel myostatin inhibitor that is optimized to block both myostatin and activin A signaling, two key regulators of muscle growth, in a balanced manner.
- Initiate taldefgrobep Phase 2 study in obesity in 2Q 2024
- Report taldefgrobep Phase 3 topline results in SMA in 2H 2024
First-in-class TRPM3 Antagonist: BHV-2100 is a first-in-class, oral, selective TRPM3 antagonist that offers a novel, non-addictive treatment for migraine and neuropathic pain. The preliminary pharmacokinetic and safety data from the ongoing Phase 1 study in healthy volunteers supports evaluation of BHV-2100 in acute migraine. It is rapidly absorbed and achieves 90% inhibitory concentrations within 1 hour, and it is well tolerated at projected therapeutic concentrations.
- Initiate BHV-2100 Phase 2 study in acute migraine in 2H 2024
- Conduct BHV-2100 POC study for neuropathic pain in 2H 2024
TYK2/JAK1 Inhibitor: BHV-8000 is a first-in-class, oral, brain-penetrant, selective TYK2/JAK1 inhibitor with broad potential for neuroinflammatory disorders. In the ongoing Phase 1 study in healthy volunteers,
- Initiate BHV-8000 Phase 2 study in Multiple Sclerosis in 2Q 2024
- Initiate BHV-8000 Phase 2a study in prevention of amyloid therapy induced ARIA in 2H 2024
- Initiate BHV-8000 Phase 2/3 study in early Parkinson's disease in 2H 2024
- Initiate BHV-8000 Phase 2/3 study in early Alzheimer's disease in 2H 2024
Extracellular protein degradation platform: Four agents quickly advancing
From Biohaven's targeted extracellular protein degradation platform, the Company is planning four INDs across a number of indications. The lead program, BHV-1300, offers a mechanism of action that is differentiated from FcRn targeting agents with the potential for a faster onset of action, deeper reductions in IgG, no mechanistic effects on albumin or cholesterol, self-administered subcutaneous dosing, and ability to dose in conjunction with Fc-containing biologic therapeutic agents. In a preclinical model, BHV-1300 demonstrated that it can be co-administered with Fc-containing biologics, such as Humira®, supporting Biohaven's strategy of advancing BHV-1300 in combination with standard of care treatments for rheumatoid arthritis (unlike the FcRn class which have limitations on co-administration with Fc containing biologics).
- BHV-1300 clinical data regarding first-in-human of IgG lowering expected in 1Q 2024
- A total of 4 INDs are expected for the degrader program in 2024
Next Generation ADC Platform:
- Initiate Phase 1 trial of BHV-1510 (Trop2) in 2Q 2024
- File IND for BHV-1500 (next gen brentuximab ADC) in 2H 2024
About Biohaven
Biohaven is a biopharmaceutical company focused on the discovery, development and commercialization of life-changing treatments in key therapeutic areas including neuroscience, immunology and oncology. The company is advancing one of the industry's most innovative therapeutic portfolios, leveraging its proven drug development experience and multiple, proprietary drug development platforms.
Forward-looking Statements
This presentation includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including statements about
Humira® is a registered trademark of AbbVie Inc.
Investor Contact:
Vice President, Investor Relations
jennifer.porcelli@biohavenpharma.com
+1 (201) 248-0741
Media Contact:
mikebeyer@sambrown.com
+1 (312) 961-2502
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/biohaven-highlights-progress-across-innovative-portfolio-and-outlines-2024-anticipated-milestones-at-the-42nd-annual-jp-morgan-healthcare-conference-established-extensive-portfolio-across-20-therapeutic-indications-in-neuroscie-302027933.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/biohaven-highlights-progress-across-innovative-portfolio-and-outlines-2024-anticipated-milestones-at-the-42nd-annual-jp-morgan-healthcare-conference-established-extensive-portfolio-across-20-therapeutic-indications-in-neuroscie-302027933.html
SOURCE
BIOHAVEN (BHVN)
| Volume | ||
| Market Cap | ||
| 52 Week High | ||
| 52 Week Low |
Minimum 15 minutes delayed. Source: LSEG